|
1/4/2023 0 Comments Cerapedics ifactor  Recently, a second Level 1 investigational device exemption (IDE) study was initiated for use in transforaminal lumbar interbody fusion. i FACTORTM and P-15TM are registered trademarks of Cerapedics, Inc., CO, USA. i-FACTOR is also supported by three additional published peer-reviewed clinical studies from international usage. These results were used to support FDA approval of i-FACTOR in this indication. Cerapedics i-Factor Peptide Enhanced Bone Graft is likely to hit the market sooner than expected, for the company just announced that an advisory committee. 
For example, study results published in the peer-reviewed journal Spine involving 319 patients who underwent anterior cervical discectomy and fusion surgery showed that i-FACTOR was superior to autograft in composite overall success one-year post-surgery. How does this product benefit the surgical team and aid in better outcomes?Ĭompleted clinical research indicates that i-FACTOR is statistically superior to autograft in overall patient success. While these products are commonly used by surgeons, the P-15 technology is supported by Level 1 human clinical data and has the potential to offer a safe, effective and economical alternative that surgeons need and that aligns with the values of healthcare systems. Other alternatives include bone morphogenetic proteins (BMPs), demineralized bone matrix (DBM), and stem cell therapies. This approach presents risks such as infection, nerve damage, bleeding, and chronic pain around the graft site. Historically, the gold standard was using a bone graft obtained from the patient’s pelvis to promote fusion (autograft). Marx, Ph.D., President and COO, Cerapedics.What sets this product apart from others available in the industry? “The next generation product we’re developing with DSM is showing tremendous potential, and will allow us to bring the bone forming potential of the P-15 technology into the lumbar spine,” said Jeffrey G. 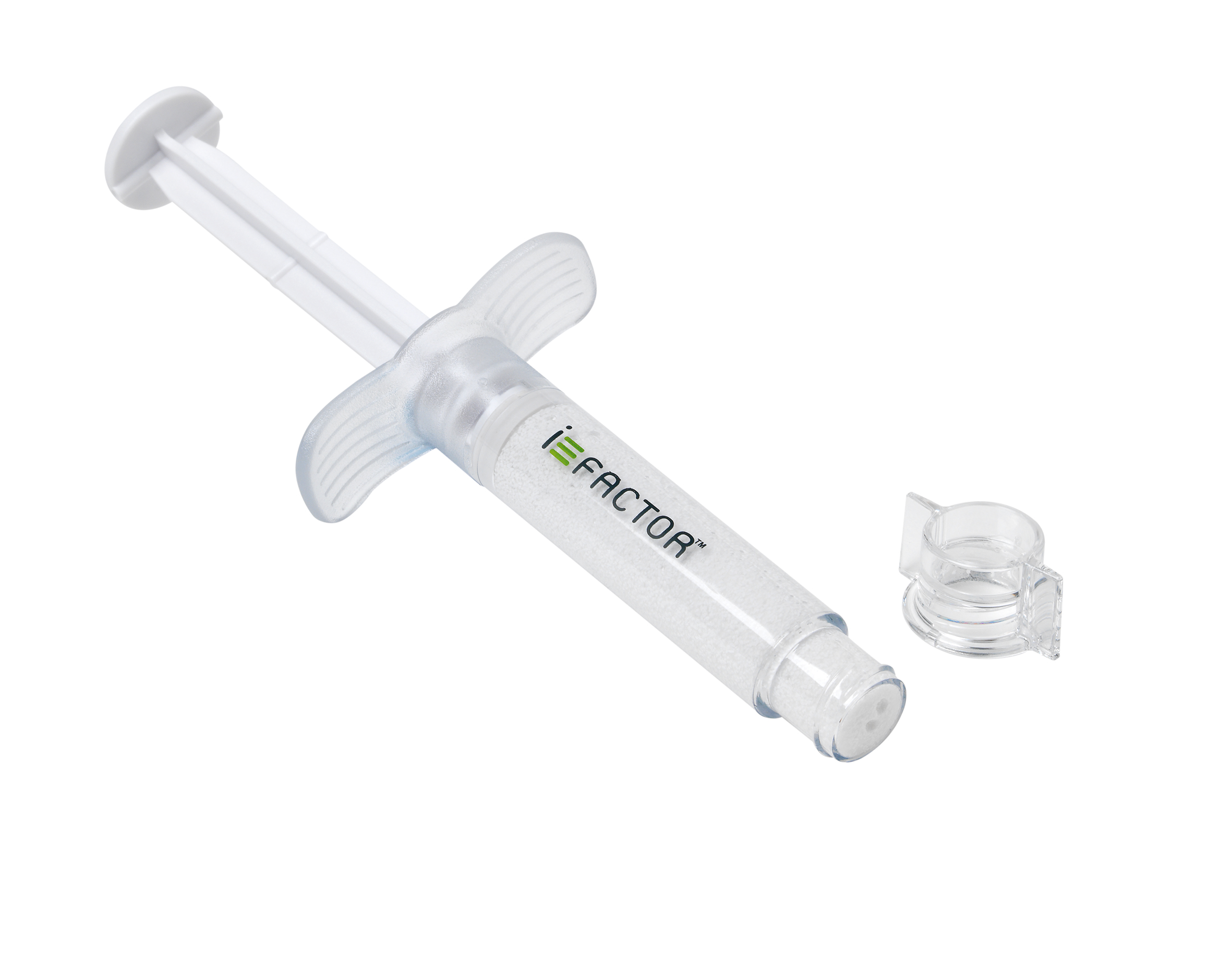
following the FDA Premarket Approval (PMA) of i-FACTOR Bone Graft, and we are excited to be partnered with DSM in delivering our next generation product to market,” said Glen Kashuba, CEO, Cerapedics. “Over the past year we have been focused on accelerating our commercialization efforts in the U.S. 
Cerapedics is partnering with DSM to collaboratively develop and commercialize the next generation of this technology. for anterior cervical discectomy and fusion (ACDF) procedures since late 2015. The company’s first-generation product, i-FACTOR™ Peptide Enhanced Bone Graft, has been commercially available outside of the U.S. “Together, from concept to commercialization, we are at the leading edge of advanced healing solutions.”Ĭerapedics developed P-15 technology to support bone growth through cell attraction, attachment, and activation. “The partnership combines the industry leading expertise and capabilities in bioceramic materials of DSM with Cerapedics’ innovative growth factor, delivering a new product solution to improve people’s lives,” said David Yonce, Vice President and Global Head of Innovation at DSM Biomedical. i-FACTOR Peptide Enhanced Bone Graft is the only biologic bone graft that uses a unique small peptide attachment factor (P15 Osteogenic Cell Binding Peptide). #CERAPEDICS IFACTOR FULL#Under the partnership agreement with Cerapedics, DSM will also provide a collagen carrier, final packaging, and full support from concept through commercialization. Among 400+ spinal bone grafts on the market, spine surgeons can. The strategic partnership aligns DSM’s regenerative materials capabilities with proprietary synthetic small peptide (P-15) technology developed by Cerapedics.ĭSM’s newest bioceramic platform is a carbonated apatite matrix designed for fast, predictable remodeling in bone graft substitute applications. Cerapedics is an advanced orthobiologics company with i-FACTOR Peptide Enhanced Bone Graft. DSM Biomedical, a global solutions provider in biomedical science and regenerative medicine, today announced that it is partnering with Cerapedics, a privately-held orthobiologics company, to develop and manufacture the next generation peptide enhanced bone graft. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
